
functional disturbances following cardiac surgery ( I97.0- I97.1).
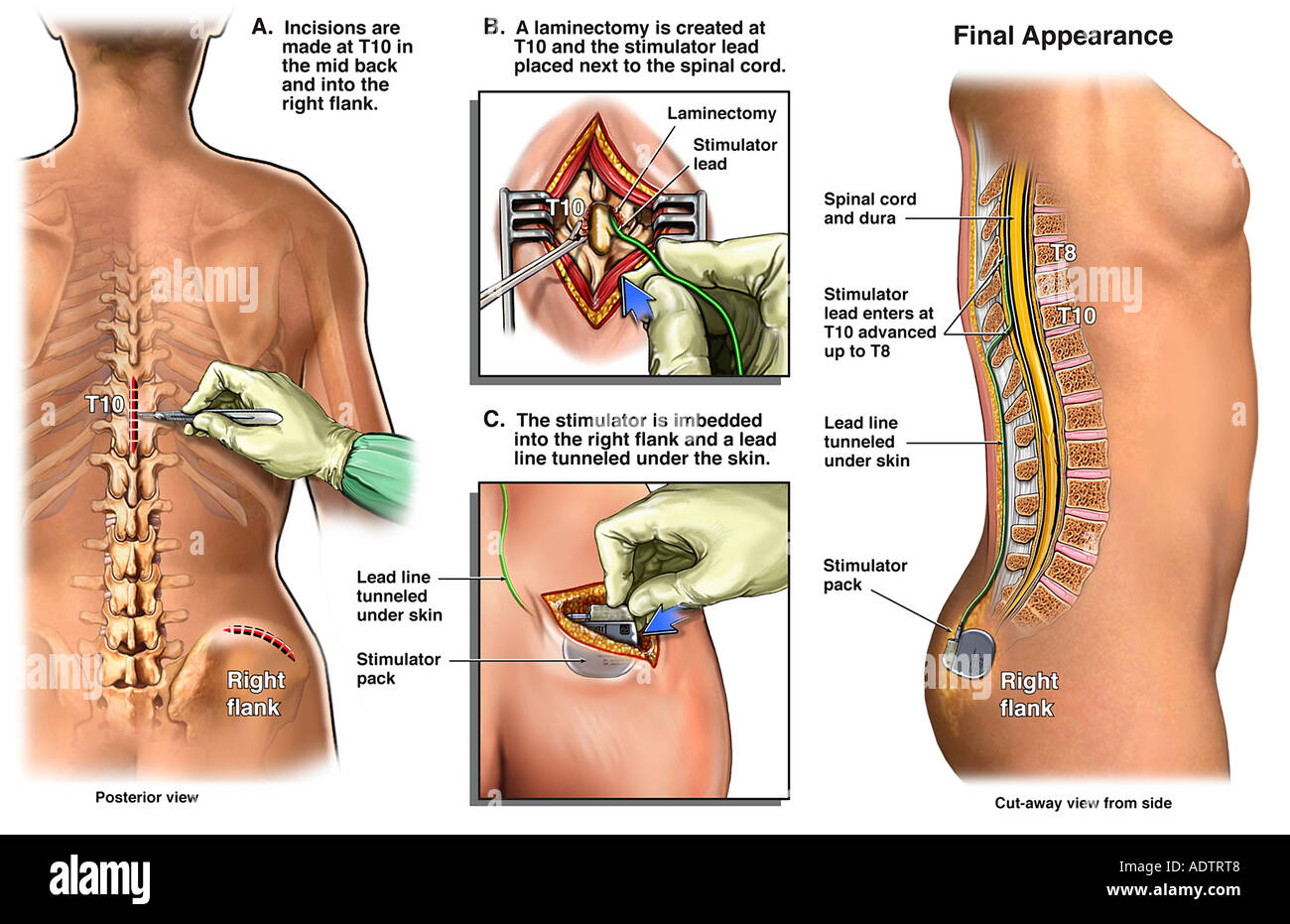
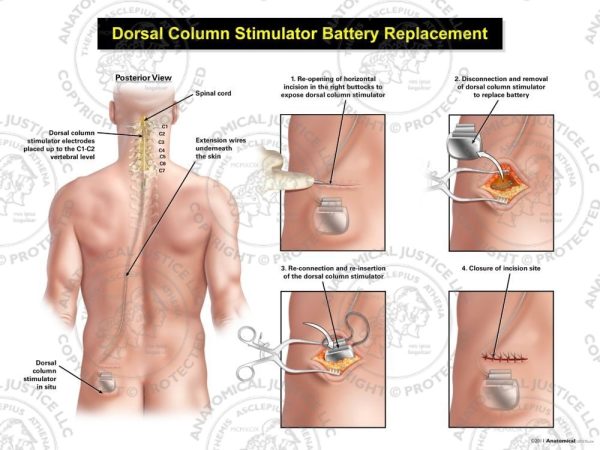
disorders of fluid and electrolyte imbalance ( E86- E87).cerebrospinal fluid leak from spinal puncture ( G97.0).specified complications classified elsewhere, such as:.poisoning and toxic effects of drugs and chemicals ( T36- T65 with fifth or sixth character 1-4 or 6).mechanical complication of respirator ( J95.850).complications of surgical procedures during pregnancy, childbirth and the puerperium ( O00-O9A).burns and corrosions from local applications and irradiation ( T20- T32).fitting and adjustment of external prosthetic device ( Z44.-).any encounters with medical care for postprocedural conditions in which no complications are present, such as:.However, in a 2020 letter to health care providers, the FDA reported that over the preceding four-year period, it received “a total of 107,728 medical device reports related to spinal cord stimulators intended for pain, including 497 associated with a patient death, 77,937 with patient injury, and 29,294 with device malfunction.” That seems like an unacceptably high rate of unintended effects for a device with modest benefits. Alan Roth and Andy Lazris noted that “Surgery for low back pain is one of the most overused procedures in the United States, with more than 1.2 million back surgeries performed every year.” At an estimated 50,000 procedures annually, spinal cord stimulators make up a relatively small portion of these back surgeries. In an American Family Physician 2022 Lown Right Care article on interventional procedures for low back pain, Drs. Food and Drug Administration (FDA) through the “substantial equivalence” 510(k) pathway with evidence of effectiveness and safety, even though this pathway does not require evidence of that kind. Finally, critics conflated approval of spinal cord stimulators by the U.S. They stated that “the credibility of our review team was attacked because one of the authors … had authored books on harms in health care,” equating this intellectual interest with financial conflicts held by supporters of spinal cord stimulators. These tactics included publishing lengthy critiques in paywalled industry-affiliated journals rather than the journal where the original study was published. In a recent commentary in JAMA Internal Medicine, two of the Cochrane review authors discussed tactics used by the spinal cord stimulator industry to dismiss the findings of their review and other independent reviews and studies that came to similar conclusions. The data do not support the use of spinal cord stimulation to manage low back pain outside of a clinical trial, and it is unclear if spinal cord stimulation has long-term clinical benefits to reasonably outweigh the costs and risks of surgical intervention. Three smaller trials suggested that “adding spinal cord stimulation to medical management may slightly improve function and slightly reduce opioid use in the medium term (i.e., one to less than 12 months).” Based on these findings, Dr. The largest study, with 50 participants, found no statistical benefits. Overall, the body of evidence was assessed as having significant bias, including selection bias (five studies), performance and detection bias (10 studies), attrition bias, and selective reporting bias. Most studies reported outcomes at follow-up dates of one month or less only one study reported outcomes at six months. Primary outcomes included pain intensity, physical function, and quality of life. Brian Nelson and colleagues summarized a Cochrane review of 13 placebo-controlled trials with 699 adult participants (mean age 47 to 59 years) who had low back pain for at least 12 weeks. These devices come with a high price tag ($30,000) and potential complications that include electrode migration, hematoma formation, infection, spinal cord injury, and cerebrospinal fluid leak. A Cochrane for Clinicians article in the December 2023 issue of American Family Physician reviewed randomized trials assessing the effectiveness of surgically implanted spinal cord stimulation devices for the treatment of chronic low back pain.
